Producing nanoparticles by contact glow discharge electrolysis

|  |
When operated under high current densities (typically larger than 1 A/mm2), gas bubbles grow dense enough on an electrode to allow the formation of a thin gas film by bubble coalescence. Electrical discharges take place between the electrode and the electrolyte.
This effect is known in the literature under various names, such a electrode effects, electrochemical discharges or glow discharge electrolysis. It was first described in 1844 by the French physicists Foucault and Fizeau. We use this effect to synthesis a large variety of metlic nano-particles
Synthesis of nano-particles by glow discharge electrolysis
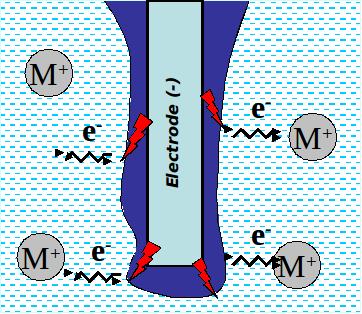
The electrical discharges taking place in the gas film formed around an electrode during glow discharge electrolysis can be used to locally reduce metal ions contained in the supporting electrolyte. The pH of the supporting electrolyte has to be chosen in order to insure the presence of the desired ions.
Particles of a large variety of metals can be synthesised, including Pt, Au, Cu, Ni. Alloys are possible as well (including Pt-Au and Ni-Cu). The size of the synthesised particles can partially be adjusted by controlling the residence time of the reactants near the electrode (rotation or using pulsed voltage).
Current Research
Our current research focuses on the following aspects:- Elucidation of reaction mechanism
- Control of size distribution of the synthesized nano-particles
- Systematic investigation of chemical and physical properties of the synthesized nano-particles
Applications
Main applications currently under investigation
- Nano-structured electocatalysts for hydrogen evolution reaction
- Nano-structured super-capacitors
- Nano-structured bifunctional electocatalysts
